XRD analysis
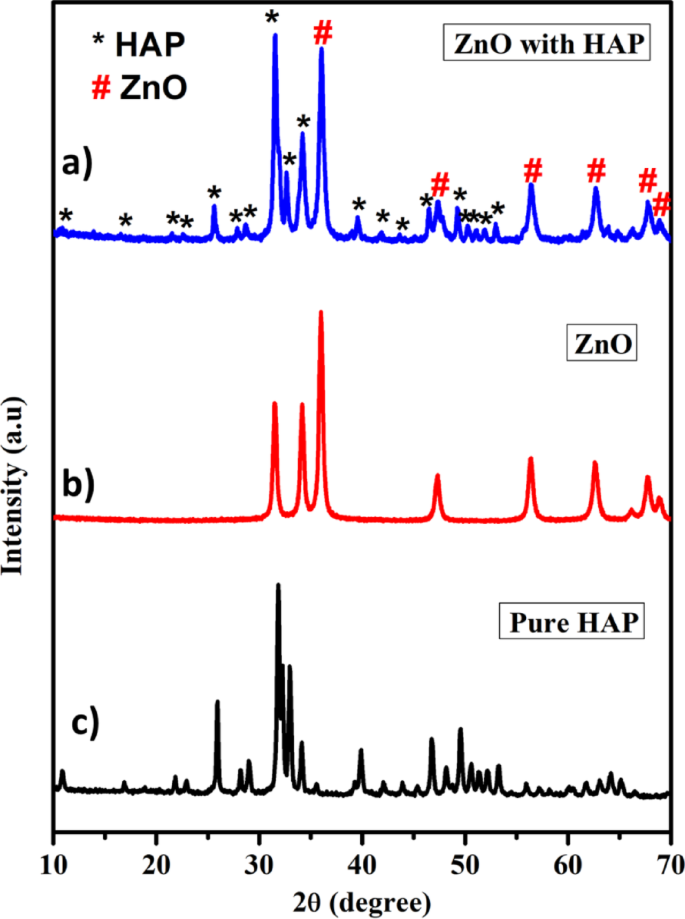
XRD patterns of photocatalysts.
The synthesized catalyst’s crystallinity, phase, and purity were investigated using an X-ray diffractometer operating in the 10–80° range with cu kα radiation (λ = 1.5418 Å). The ZnO@HAp composite, zinc oxide nanoparticle, and pure HAp’s XRD patterns are displayed in Fig. 1a–c, respectively. Figure 1a shows that all of the diffraction peaks were in good agreement with the pure hexagonal phase of hydroxyapatite (HAp). Figure 1b demonstrates that the diffraction peaks were more intense and narrower in ZnO, and this observation shows that the prepared ZnO has a wurtzite phase with a good crystalline nature10. Figure 1c shows the XRD pattern displaying characteristic peaks of the hexagonal phase, corresponding to the space group P63/m (ICDD 00-009-0432 file). The distinct presence of HAp is confirmed by the significant diffraction peaks seen at the (211), (112), and (300) planes. These peaks remained stable in the nanocomposite even after ZnO impregnation. This suggests that in the prepared samples, HAp is the predominant phase. Furthermore, ZnO with a hexagonal crystal structure, which belongs to the space group P63/mc (JCPDS card no. 89-1397), is detected by the XRD pattern24. The ZnO@HAp composite’s diffraction patterns remained constant even after it formed, indicating that the structure of the materials is sustained in the composite. The peaks at 2θ: 36.36°, 56.7°, 63.02°, and 68.09° positions correspond to zinc oxide hybridized with hydroxyapatite to form a nanocomposite24. The XRD pattern revealed that the result was a pure ZnO@HAp composite, as no contaminants were present in the particles.
FTIR analysis
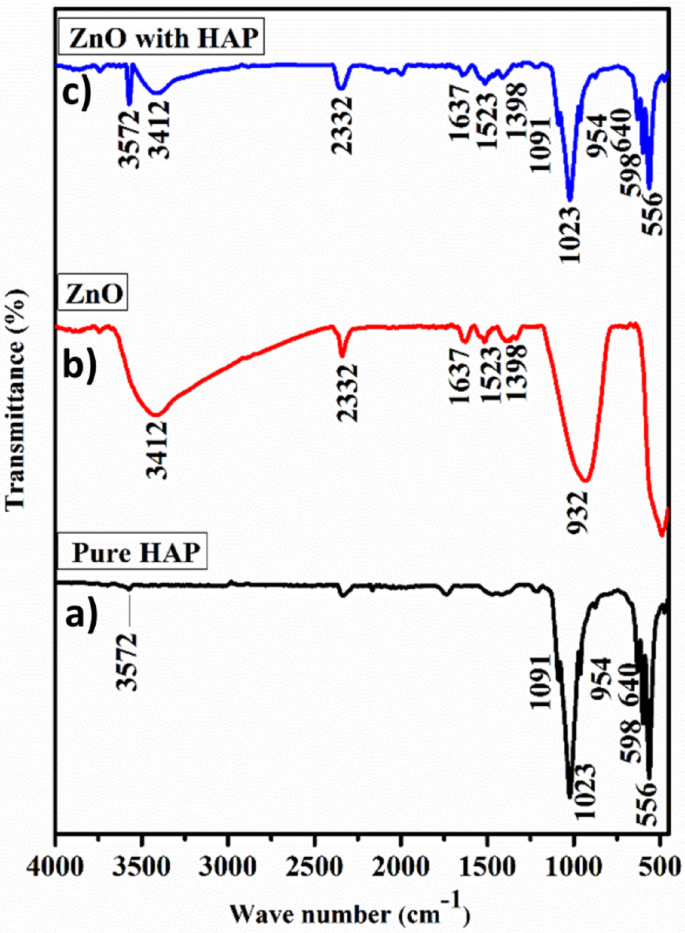
FT-IR results of photocatalysts.
Figure 2a–c presents the FT-IR spectra of the pure HAp, ZnO, and ZnO@HAp composite. The peaks at 598 and 556 cm− 1 in the FT-IR spectra of hydroxyapatite nanoparticles (Fig. 2a) are linked to O–P–O vibrations and represent the triply degenerate bending modes of the phosphate (PO43−) group. The peaks at 1023 and 1091 cm− 1 are ascribed to the P–O bond’s asymmetric stretching vibrations. Furthermore, the vibrations of the hydroxyl (–OH) group found in HAp are related to the peaks observed at 3572 and 640 cm− 125. The peaks located at 533 and 3412 cm− 1 in the case of zinc oxide nanoparticles are associated with the stretching vibrations of the Zn-O bond and −OH. Both the distinctive peaks of zinc oxide and hydroxyapatite nanoparticles distinctive peaks were visible in the ZnO@HAp composite’s spectrum, especially at 556, 640, 1023, 2081, and 3572 cm− 1, which validated the composite’s formation (Fig. 2c).
Scanning electron microscopy
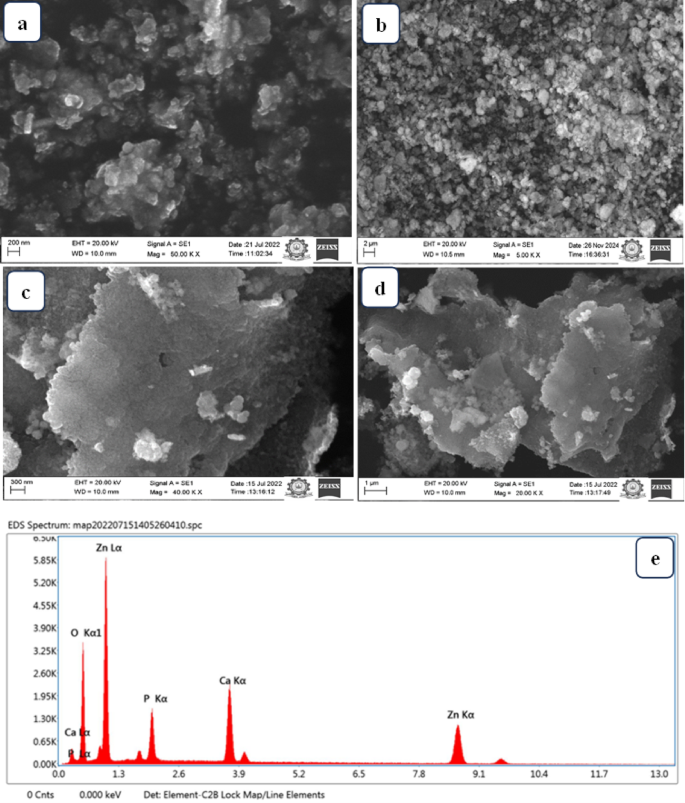
SEM-Morphological structure of (a) ZnO, (b) HAp, (c,d) ZnO@HAp, and (e) EDAX data of ZnO/HAp.
The SEM images were used to analyze the HAp crystal’s surface morphology. The results shown in Fig. 3a indicate that ZnO particles exhibit regular shapes with approximate sizes of 20–50 nm, and they are also agglomerated. The particle size distribution and different morphologies of platelets and flaky particles were predominant. The HAp sample’s SEM images are displayed in Fig. 3b. It illustrates the HAp’s general morphology. The HAp powder contains aggregates and granular surfaces, according to the SEM analysis. SEM images of the ZnO@HAp at two magnifications are depicted in Fig. 3c,d. We could see that the particles were agglomerated, and spherical-shaped ZnO particles were present on the surface of HAp. EDAX data of prepared material ZnO@HAp, as seen in Fig. 3e, indicate the presence of zinc, oxygen, phosphorus, and calcium in the prepared materials, confirming the formation of the composite.
BET analysis
The developed photocatalyst’s nitrogen adsorption and desorption isotherms are shown in Fig. 4. The presence of porous materials is indicated by the type-IV nitrogen adsorption-desorption isotherms with a noticeable hysteresis loop in the relative pressure (P/Po) range of 0.2 to 1.0, which are displayed by both ZnO and ZnO@HAp. In comparison to pure ZnO, the ZnO@HAp nanocomposite’s BET study showed a noticeably larger surface area. With a total pore volume of 0.0929 cm3/g, micropore volume of 0.004 cm3/g, micropore surface area of 9.179 m2/g, and exterior surface area of 87.096 m2/g, the calculated specific surface area was 110.124 m2/g. These findings demonstrate that ZnO@HAp has a significantly higher BET surface area (110.124 m2/g) than pure ZnO (30.2 m2/g) and a well-developed porous structure. The higher surface area of ZnO@HAp enhances its adsorption characteristics, consequently increasing photocatalytic activity.
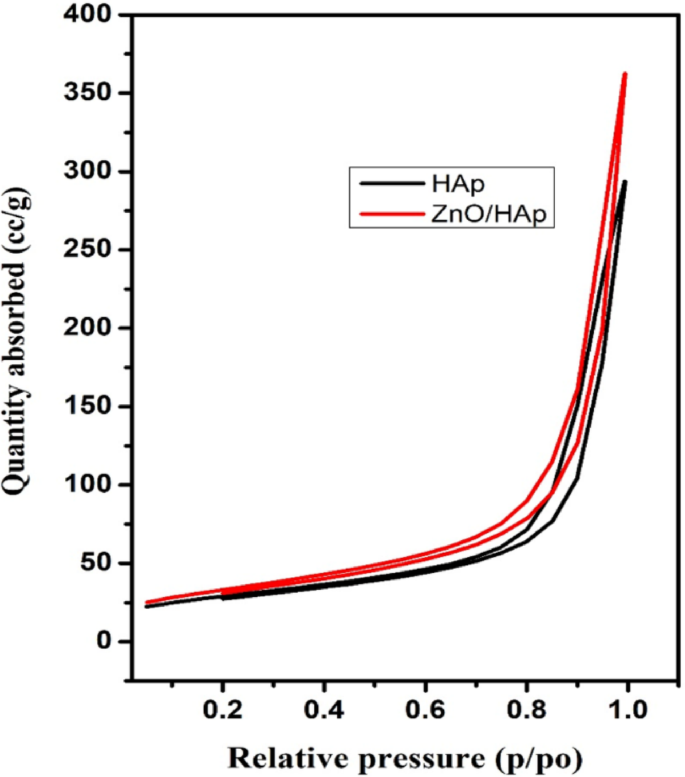
Nitrogen adsorption isotherms of the prepared materials.
TEM analysis
TEM research was used to further investigate the synthesized photocatalyst’s structural and morphological properties. The presence of particles in various forms, including spherical and hexagonal structures of pure HAp, is visible in the TEM images of the material (Fig. 5a,b). The findings of the SEM are in agreement with these observations. Moreover, ZnO@HAp nanoparticles are comparatively monodispersed, with an average particle size ranging from roughly 25 to 40 nm, according to the TEM images of ZnO@HAp nanocomposites (Fig. 5c,d).
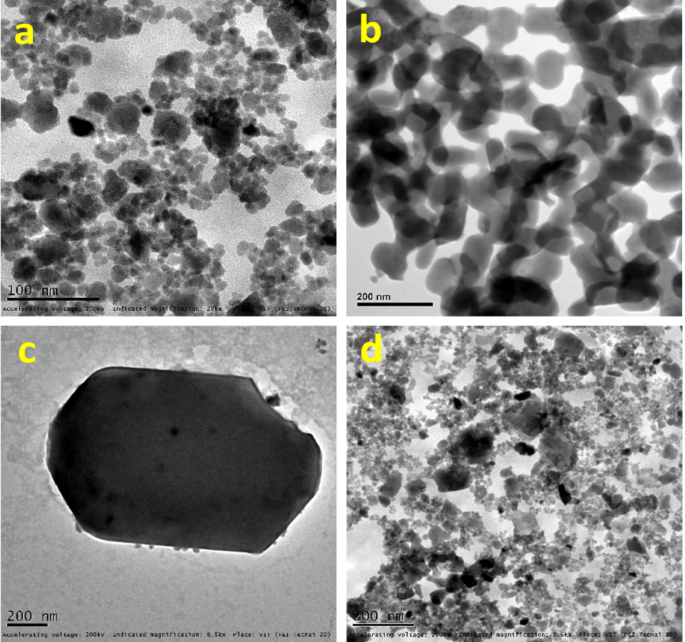
TEM images of HAp (a,b) and ZnO@HAp (c,d).
XPS analysis
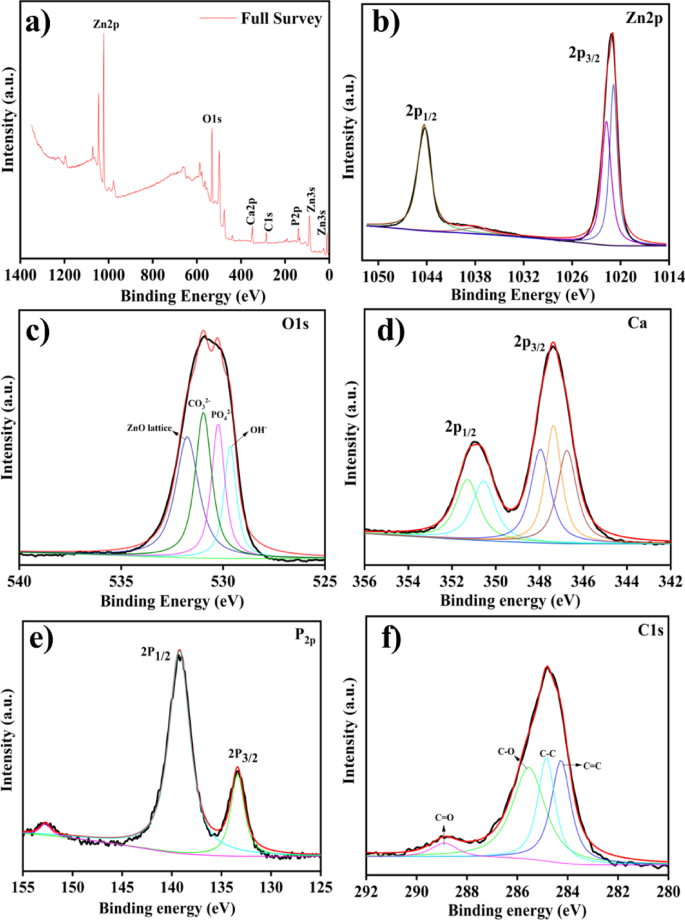
XPS spectra of ZnO/HAp (a) Full survey, (b) Zn 2p, (c) O 1s, (d) Ca 2p, (e) P 2p, and (f) C 1s.
X-ray Photoelectron Spectroscopy (XPS) was performed to investigate the surface chemical composition of the as-synthesized ZnO@HAp composite. Figure 6 displays the binding energies corresponding to the elemental states of O, Ca, P, Zn, and C. The survey spectrum (Fig. 6a) confirms the presence of Zn, O, Ca, P, and C through their characteristic peaks. In particular, the Zn 2p region (Fig. 6b) shows two distinct peaks at 1021.7 eV and 1044.1 eV, which correspond to Zn 2p3/2 and Zn 2p1/2, respectively, indicating the formation of Zn–O bonds in the composite26. The O 1s spectrum (Fig. 6c) was deconvoluted into four distinct peaks located at 529.6 eV, 530.2 eV, 530.9 eV, and 531.7 eV. These peaks can be attributed to lattice oxygen in ZnO, carbonate (CO32−), phosphate (PO43−), and hydroxyl groups (OH−), respectively27. Figure 6d shows two major peaks at 347.9 eV and 351.3 eV, corresponding to the Ca 2p3/2 and Ca 2p1/2 core levels, which confirm the presence of calcium in the composite. The Ca 2p3/2 peak originated from calcium bonding and its interaction with adsorbed CO32− in HAp, while the Ca 2p1/2 peak corresponded to metallic calcium28. The P 2p spectrum (Fig. 6e) displays two significant peaks at 133.3 eV and 139.2 eV, assigned to P 2p3/2 and P 2p1/2, respectively, indicating the presence of P–O bonds in the HAp structure29. Finally, Fig. 6f presents the C 1s and ZnO region of the XPS spectrum. The C 1s peak appears at 284.8 eV and is typically used as a reference for charge correction. Notably, the binding energy of the C 1s peak remains unchanged after the partial substitution of Ca2+ ions with Zn2+ ions, and consistent with previous reports30.
Photocatalytic activity and effect of catalyst amount
The photocatalytic degradation of methylene blue(MB) dye under UV irradiation (365 nm) was investigated using ZnO and ZnO@HAp catalysts. Even after 30 min, we don’t see the complete degradation of methylene blue dye. We have achieved a degradation efficiency of less than 40%. Hence, the ZnO alone may not be useful for industrial dye degradation. In general, HAp does not degrade organic dye due to its large energy band gap, which might be the reason why the photocatalytic efficiency has not been improved over HAp. As seen in Fig. 7a, the methylene blue undergoes almost90% degradation in 5 min with ZnO@HAp.
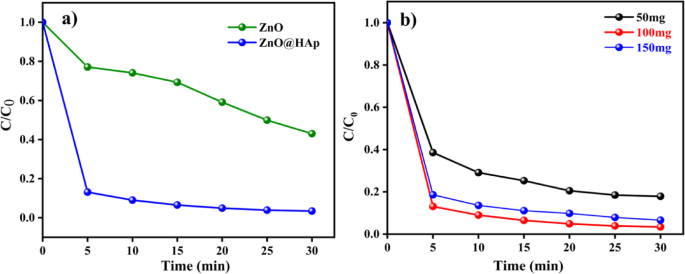
(a) Methylene blue (10 ppm/50 mL) degradation with ZnO, ZnO@HAp (50 mg), pH 7, and (b) catalyst dosage of ZnO@Hapon MB degradation.
Photocatalytic studies were conducted using varying doses of the catalyst, ranging from 50 to 150 mg, to determine the optimal dosage of ZnO@HAp. Figure 7b illustrates how the availability of additional active sites and higher production of hydroxyl radicals caused the degradation rate of methylene blue to increase with catalyst loading up to 100 mg. However, efficiency decreased when the catalyst amount exceeded 100 mg. This was probably because increased turbidity and light scattering made it more difficult for photons to penetrate. Therefore, 100 mg was found to be the optimal dosage of ZnO@HAp for efficient methylene blue degradation. As shown in Fig. 8a, the ZnO@HAp (k = 0.0281 min− 1) catalyst exhibits a pseudo-first-order rate for MB degradation that is higher than pure ZnO (k = 0.1127 min− 1), indicating enhanced photocatalytic efficiency. Error bars (Fig. 8b) represent standard deviations from triplicate experiments, confirming the reliability of the degradation performance.
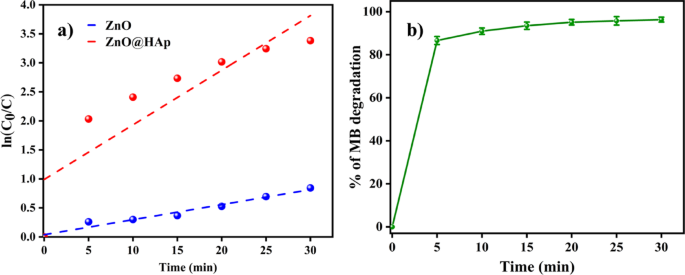
(a) Methylene blue degradation kinetics of ZnO, ZnO@HAp, and 0 (b) Error bar of ZnO@Hapon MB degradation.
The absorption spectra data of methylene blue over ZnO@HAp with different irradiation time intervals are shown in Fig. 9a. The optical energy band gap of ZnO@HAp, calculated using a Tauc plot, was found to be 3.15 eV (Fig. 9b), which is lower than the bandgap energies reported for ZnO (3.26) and HAp (4.2 eV)31,32. The optical features of ZnO@Hap nanocomposite cause charge transfer from ZnO to HAp and reduce charge carrier recombination in zinc oxide. As a result, hydroxyl radicals are produced on composite surfaces, accelerating the photodegradation process. The ZnO@HAp photocatalyst is found to be much more efficient than ZnO and HAp. The higher efficiency of ZnO@HAp might be due to the strong interaction between ZnO and HAp. Additionally, the SEM analysis reveals that ZnO nanoparticles are located on the surfaces and within the pores of the HAp. This could be responsible for the increased catalytic activity of the ZnO@HAp composite. No degradation was observed when the reaction proceeded without a catalyst. Therefore, compared to pure ZnO, ZnO@HAp’s greater specific surface area and porous architecture improve its photocatalytic efficacy toward organic pollutants. Results confirm that ZnO was evenly combined with the HAp matrix in the ZnO@HAp nanocomposite, which exhibits a high specific surface area relative to individual ZnO and HAp. The amount of the catalyst and recyclability are important aspects of photodegradation.
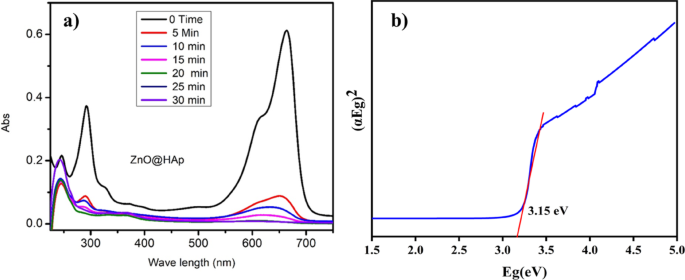
(a) Methylene blue (10 ppm/50 mL) degradation absorption spectra with ZnO@HAp (50 mg), pH 7, and (b) Tauc plot of prepared ZnO@HAp.
The pH of the solution plays a crucial role in the photocatalytic degradation process. Investigating the effect of pH in the range of 4–9 (Fig. 10) revealed that the maximum degradation efficiency of 96.4% was achieved at a neutral pH of 7 within 30 min. The reduced effectiveness below this pH is attributed to the partial solubility of ZnO in the ZnO@HApcatalyst under acidic environments.
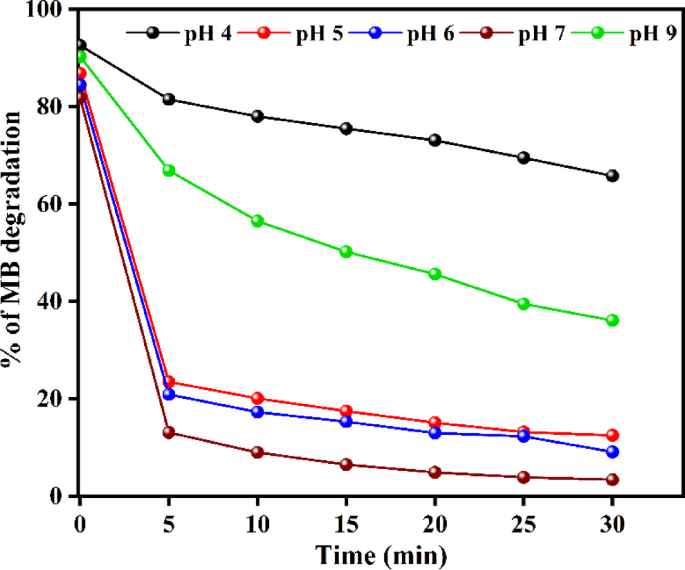
Effect of initial solution pH on MB degradation.
Since the catalyst surface becomes more negatively charged and the electrostatic affinity with anionic dye species is weakened, the degradation efficiency decreases above pH 7, resulting from reduced adsorption of dye molecules. For pH levels of 4, 5, 6, 7, and 9, the corresponding adsorption measurements at equilibrium were 7.4%, 13.2%, 15.6%, 19.1%, and 9.7%, respectively. The highest adsorption was observed at a pH of 7. The ideal pH for methylene blue mineralization is 7, as confirmed by the strong correlation between adsorption and degradation efficiency.
Catalyst recyclability
Recycling studies of ZnO@HAp for the degradation of methylene blue dye under optimized reaction conditions were performed over 5 cycles, and the results are shown in Fig. 11a. It is important to note that even after the five consecutive recycles, the photocatalytic efficiency is not much affected. In addition, the recycled catalyst was separated and dried, and then analyzed by powder XRD. The XRD pattern of the recycled photocatalyst is shown in Figure S1 (Please see supporting information). We found no significant changes even after five recycles. These results clearly indicate the stable nature of the catalyst.
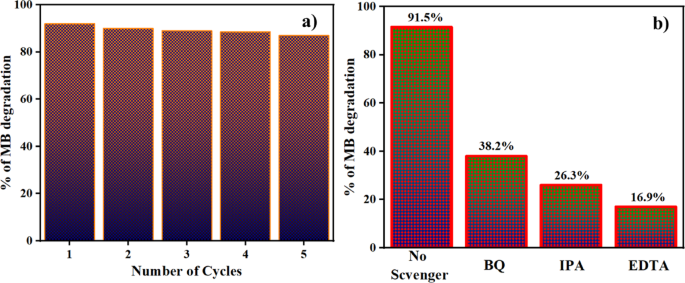
(a) Reusability of ZnO@HAp, and (b) Scavenger test of ZnO@HAp catalyst.
Scavenger test
The process of photocatalytic degradation occurs through the generation of radicals with light. If radical scavengers are introduced, they can reduce the concentration of radicals and impact the rate of photodegradation. To identify the active radical species responsible for photocatalytic activity, four scavengers were used during the degradation of the methylene blue dye. The ethylenediaminetetraacetic acid (EDTA, 10 ppm), p-benzoquinone (BQ, 10 ppm), isopropyl alcohol (IPA, 10 ppm), and ethanol were used as h+, superoxide radical (·O2−), and hydroxyl radical (·OH) scavengers, respectively. The results indicate that the EDTA scavenger, which removes holes (h+) from the system, has a significant impact on the assessment of photocatalytic activity (Fig. 11b). As per the scavenger test, holes (h+) are considered the principal active species in photocatalytic degradation. As shown in Fig. 11b, the addition of BQ caused the degrading efficiency to decrease moderately by 53%, while the addition of IPA and EDTA, at the same volume and concentration, caused a considerable suppression of roughly 65% and 75%, respectively. Therefore, hydroxyl radicals (·OH) and holes (h+)play the dominant role in the photodegradation of methylene blue; however, h⁺ plays a more significant role in the ZnO@HAp photocatalytic system than ·OH. Additionally, the efficiency of the catalyst was investigated for the degradation of the drug ciprofloxacin and in the treatment of domestic wastewater. Ciprofloxacin degradation is shown in Fig. 12a. About 63% of degradation occurred in 150 min of irradiation. The kinetic plot for ciprofloxacin degradation is shown in Figure S2 (Please see supporting information). In domestic wastewater treatment, both COD and TDS were measured to monitor the degradation process. Changes in COD and TDS are displayed in Fig. 12b. About 90% COD reduction was observed in 150 min of irradiation. These results confirm the efficiency of the catalyst in the treatment of domestic and pharmaceutical industrial wastewater.
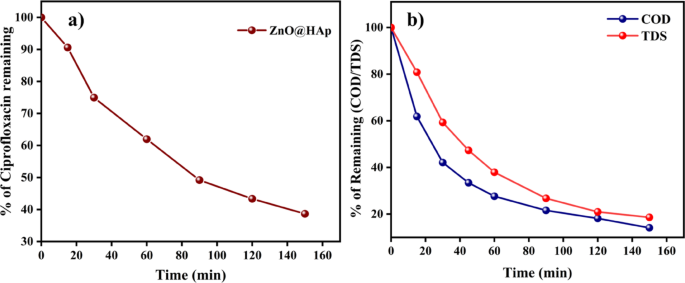
Ciprofloxacin degradation (a) Sludge wastewater treatment of ZnO@HAp (b).
Based on the scavenger test, a proposed mechanism of degradation is shown in Fig. 13. The concentration of hydroxyl groups on the catalyst surface is significantly increased by the addition of HAp to ZnO. Pollutant breakdown is facilitated by the increased number of hydroxyl groups, which act as efficient trapping sites for photogenerated holes33. Additionally, the presence of HAp increases the number of molecular oxygen adsorption sites, facilitating the trapping of photogenerated electrons and producing more hydroxyl radicals. Furthermore, the high adsorption characteristics of HAp enable it to adsorb the pollutant molecules and release them synergistically for degradation by ZnO. Higher surface area of ZnO@HAp (110.124 m2/g) than pure ZnO (30.2 m2/g causes an increased adsorption of pollutants and therefore, as compared to pure ZnO, the ZnO@HAp nanocomposite demonstrated better photocatalytic activity.
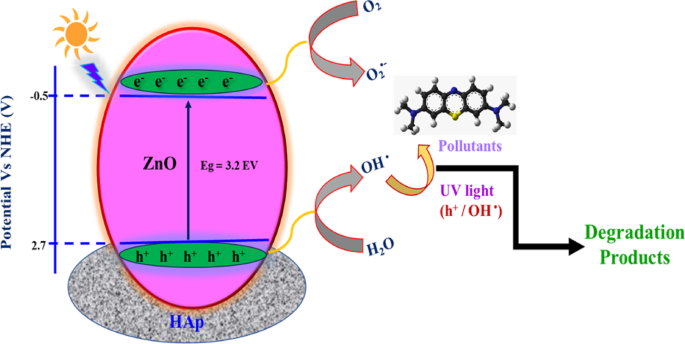
Degradation mechanism of ZnO@HApcatalyst.
The photocatalytic performance of ZnO@HAp for MB degradation is compared with earlier results in Table 1 to demonstrate its effectiveness. ZnO@HAp demonstrated higher activity and reusability, achieving 96% degradation in 30 min and maintaining 87% efficiency after five cycles, whereas many other reported catalysts required longer timeframes34,35,36,37.


3 Comments
https://shorturl.fm/DkMsT
https://shorturl.fm/Fvaj3
https://shorturl.fm/LWgeg